Ten years ago, if you told me that I was going to dye my hair red when I was 21 years old, I would have told you that you were absolutely lying! I would never (according to 11-year-old me). But according to 22 year old me, yes — I absolutely did dye my hair red, and it’s here to stay for the next few years because I absolutely love it!
The first time I dyed my hair, it was in the Fall of 2022. I wanted a big change (for personal reasons I will not delve into), but I wanted to do something I never did before. Something that will make me feel new. It did not come for cheap, however. It was worth it nonetheless. This lovely thing I’m talking about is hair dyeing!!
So, what exactly goes behind hair dyeing? It all boils down to chemistry! The process is different depending on the type of hair dye you get: temporary/semi-permanent, demi-permanent, and permanent. Since I decided to get permanent red hair dye, I will discuss about the permanent hair dye process. Permanent color is what is needed for a more significant hair color change (i.e. from black to red or blond).
Your natural hair color depends on the quantities and ratios of 2 main proteins: phaeomelanin and eumelanin. Eumelanin causes hair to be brown or black while phaeomelanin causes hair to be blond and red shades. My hair is jet black, so that means my hair is mostly composed of eumelanin.

Both ammonia and hydrogen peroxide are used. Ammonia and peroxide act to lighten the hair’s natural color to form a new base color that will allow the permanent hair dye to stay.
The cuticle, or outer layer, or the hair strand must be opened before permanent hair color can be applied into the hair. Once the cuticle is open, the dye reacts with the cortex of the hair to deposit or remove color. The cortex is the part of the hair that is the middle layer of the hair shaft which provides the strength, color and texture of a hair strand. There is usually a two-step process for permanent hair coloring: first to remove the original color of the hair, and then the second to apply a new color.
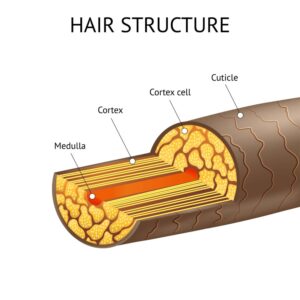
The alkaline chemical of ammonia is used to open the cuticle to allow the hair color to enter the cortex of the hair. Additionally, it is a catalyst when the permanent hair color combines with the peroxide. Hydrogen peroxide is used as an oxidizing agent, or a developer to remove pre-existing color (it removes the melanin in the hair). Peroxide breaks chemical bonds in the hair, which releases sulfur. This is why there is such a strong odor when dying hair. As the melanin is removed from the hair, the new permanent color is bonded to the cortex of the hair.

And voila! The hair dresser then washes your hair and styles it. I opted for a haircut and a blowout hairstyle. Shoutout to my talented hairdresser Christi at Cherry Blossom Salon!
Have you ever wondered what dyed hair vs non-dyed hair looks like under a microscope?
Well look no further! With the help of Dr. Leavey, a classmate and I were able to look under the microscope of the molecules of our hair. The picture on the left is mine (red dyed hair) and the one on the right is my peer Claire’s hair (undyed black hair). As you can see, my hair cuticle and cortex are much more jagged and rough, compared to Claire’s smooth strand. This is a result of the chemical reactions that occurred to my hair – the ammonia and peroxide chemically and physically damaged my hair to allow the red color to deposit into my strands.


What is wild is that my dyed hair is essentially permanently damaged. I will have to grow out my original black hair if I want to revert back to my undamaged hair. For now, I think I will stick with damaged dyed hair! 🙂
Sources:
Anne Marie Helmenstine, P. D. (2019, July 14). The science behind how hair coloring works. ThoughtCo. Retrieved April 26, 2023, from https://www.thoughtco.com/salon-hair-color-chemistry-602183
Brain, M. (2023, March 8). How hair coloring works. HowStuffWorks Science. Retrieved April 26, 2023, from https://science.howstuffworks.com/innovation/everyday-innovations/hair-coloring3.htm
