Recently, our STEMComm class put on a Sober Science Speakeasy event for Atlanta Science Festival, where our classmates explored the science of drinks. One of our drinks was the science behind the tastes and textures of milk tea boba, which I have depicted in a series of illustrations.
Recently, our STEMComm class put on a Sober Science Speakeasy event for Atlanta Science Festival, where our classmates explored the science of drinks. One of our drinks was the science behind the tastes and textures of milk tea boba, which I have depicted in a series of illustrations.
Milk tea is typically made with green tea or black tea.
 Teas contain polyphenols, which is the chemical responsible for the bitterness of tea.
Teas contain polyphenols, which is the chemical responsible for the bitterness of tea.
Milk contains casein proteins. When milk is mixed with tea, these proteins form a complex in which the polyphenol is encapsulated in the protein, decreasing the bitter taste.
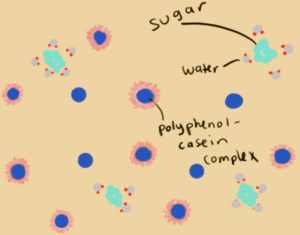
Added sugars contribute further to the sweetness of the tea as they are easily absorbed in the mouth. These sugars also bond with water molecules to leave room for the casein proteins to form complexes with the polyphenols.
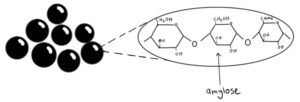
The tapioca pearls are made of amylose and amylopectin (linear and branched starch molecules). When the boba is boiled, they expand, and the glycosidic linkages (the bonds that join these molecules) create a network that is responsible for the chewy texture of the boba.
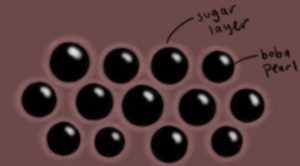
After the boba is cooked, it must be served within a few hours, or else it will harden up again. We place the boba into a solution of 1 part brown sugar, 1 part water. The sugar creates a protective layer around the boba that prolongs the hardening.
If you want to make your own boba at home, you can easily buy your own bags of dehydrated tapioca pearls on amazon or at a local asian market. Stir them into some black tea and milk with added sugar or other flavors, and voila, you have your own bubble tea!
Sources:
Dutfield, S. (2019). The science behind bubble tea. Retrieved 20 April 2020, from https://www.howitworksdaily.com/the-science-behind-bubble-tea/
Bubble Tea Infographic. Retrieved 20 April 2020, from https://www.clayton.edu/chemistry-physics/News/Infographic-bubble_tea/