I’m willing to bet you’ve never heard of Organoids. No worries if so, they’re a pretty niche part of biomedical sciences. This is such a shame however as they’re having a great impact on research and our world. Organoids are often defined as tissue samples embedded into 3D extracellular matrix (ECM) gels or clonal derivatives of cells grown without mesenchyme and derived from stem cells. Don’t worry if your eyes have already glazed over! Mine did too the first time I read that.
In simpler terms, they are the growing of cells, taken from a patient, in a nutrient rich gel. While the field might seem niche at first, it has a long history and the potential to impact millions.
Before Organoids, most human physiological research relied on in vitro 2D cell culture and animal models (O’Connell & Winter, 2019). These often fail at clinical trials due to oversimplification of or incompatibility with human systems. At the time, cell cultures were limited to growing single layers of cells in a petri dish with none of the 3D complexities of real biological systems. This all changed with Ross Harrison’s “Hanging drop Cell Cultures” in 1906 (Simian and Bissell, 2016). By creating a 2D cell culture and turning the petri dish upside down, gravity would cause the cells to clump together and form a cohesive 3D spheroid of cells that could intermingle and interact like normal. Think about that! 3D structures could form by simply turning them upside down with no other interference. This was the start of research into organoids which offered a new way to look directly at human biology without risking patient or animal lives.
One key breakthrough was the discovery of 3D extracellular matrices first used in 1963 (Simian and Bissell, 2016). The gel provided a medium for natural scaffolding of cells rather than relying on gravity. This allowed for more complex 3D shapes and allowed for more types of organoids that more closely resembled complex real life systems. The gel also aided in providing the cells with nutrients required to keep them alive. Hence the “nutrient rich gel” from the definition above.
Exploration into this new field was rather slow however. The human body is composed of roughly 200 distinct types of cells, each of which specialize in a different function such as blood or nerve cells. Despite this, organoids mainly rely on only two types of cells, pluripotent stem cells and organ-restricted adult stem cells (Bredenoord et al., 2017). Stem cells are unspecified types of cells, yet to differentiate, with the ability to become any type of specified cell. They often serve to repair the human body as they can replace any damaged cells no matter the type. This ability makes them highly valuable to organoids trying to model systems that rely on the cooperation of multiple cell types.
Before the discovery of induced stem cells in 2006 by Dr. Shinya Yamanaka at Kyoto University, organoids were made from pluripotent stem cells as organ-restricted adult stem cells can be hard to access and are restricted to becoming cell types of their tissue of origin most commonly found in embryos (Agarwal et al., 2024). The line of when a zygote becomes a person, however, is blurry, raising many ethical concerns about the desecration of a person’s body. Induced stem cells allow for the change of specialized cells back into stem cells. Organoids can now be created from any tissue sample, entirely limiting the need for embryonic stem cells outside of studying early organ development (Frans Schutgens & Clevers, 2019).
With better ways to develop organoids, scientists quickly realized their potential. Organoids offer unparalleled advancement in disease research and treatment, personalized medicine, and so much more that we simply don’t have time for here.
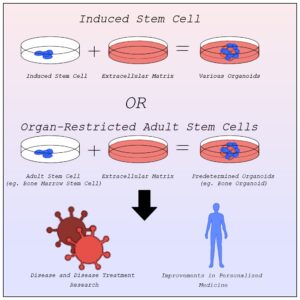
Disease Research and Treatment
One pivotal area of applications is understanding the development of diseases and disease treatment in humans. The lack of 3D structure in in vitro models lead to limited complexity and physiological relevance when studying diseases. Faulty predictions based on these studies can lead to treatments failing, with approximately 90% of clinical study drug candidates failing clinical trials and drug approval (Sun et al., 2022).
Organoid models have none of these limitations. In 2013, the first human condition modeled using organoids, cystic fibrosis, was published. The model had “excellent predictive value” and became the first successful personalized treatment method for cystic fibrosis patients (Lancaster and Huch, 2019). The modeling of other diseases using organoids quickly followed from genetic diseases like Polycystic kidney disease and Alagille syndrome, to infectious diseases like Covid-19 and the Zika virus, to neurological diseases like Alzheimer’s and Parkinson’s, to multitudes of cancers, and more (Xu et al., 2018). The ability for organoids to be made of any human system means they can model any disease regardless of which systems they afflict.
Organoids can be also grown co-culturally with bacteria allowing for bacterial infections and the diseases that arise from them to also be modeled and researched with organoids (Schutgens and Clevers, 2020). This has allowed for better understanding of bacteria like Helicobacter Pylori and Salmonella, aiding in detection and cures. As mentioned above, treatments can then be tested on these organoid systems without risking human lives or animal cruelty.
Respiratory Syncytial Virus (RSV), a disease that is being modeled in airway organoids, causes around 66,000–199,000 deaths among children annually (Schutgens & Clevers, 2020). This is just one of the many diseases for which scientists are now better equipped to find viable treatments. Organoids have already saved millions of lives and their potential for future disease treatment has the potential to millions more.
Personalized Medicine
While these treatments work for many people, genetic and biological differences can impact treatment effectiveness and tolerance with some even leading to negative side effects (Breen, 2021). In such situations, personalized medicine that is specialized to fit these differences might be necessary. Personalized medicine, sometimes called precision medicine, challenges the idea that treatments that pass general trials will work for everyone, instead working to construct a treatment plan with a specific patient in mind best suited to their physiology.
The idea of personalized medicine has been around for thousands of years, using one’s family’s history of diseases to diagnose the likelihood of that person having said disease. In the late 1900s the idea of evidence based medicine, the implementation of explicit reasoning based on a specific patient’s data, started being common practice when prescribing a patient (Visvikis-Siest et al., 2020). This opened the door for personalized medicine in modern science (Visvikis-Siest et al., 2020). This data was largely based on patients’ reactions to other treatments and family history however limited its effectiveness. The development of molecular biology and human genome mapping allows for a direct look at a patient’s physiology, aiding medical professionals in determining the biological reasons behind potential reactions to treatments before administration limiting adverse reactions.
Organoids have greatly furthered this field. As mentioned above, organoids are made from a patient’s induced stem cells. This means that the organoid culture into a structure based on the patient’s DNA, containing all the same biomarkers and physiology that the patient would have at that level. Where other treatments could have varying effects from the tested population, organoids allow medical professionals to see the exact effects a treatment plan would have on the patient. The use of stem cells also allows organoids to grow much quicker than other models. This allows for quick and accurate testing of the effects of medication on a patient. One study published in Science involving gastrointestinal cancer therapies found that organoids would correctly detect when drugs would fail 100% of the time, and when the drug would succeed 88% percent of the time (Vlachogiannis et al., 2018). Once again, because of the diversity of systems that organoids can model, organoids can be used to model many different personalized medicine plans allowing for the variety of treatment testing needed to find the most optimal plan.
Conclusion
Organoids have had a monumental, yet widely unknown, impact on a wide variety of disease advancement and treatment developments and personalized medicine. These structures can then be used to model human tissues and organs with much higher accuracy and complexity than previously used non-human or in vitro models.
Scientists use them to model disease development in humans and treatments designed for humans. This allows for a better understanding of the diseases and more effective treatments with fewer errors. They are also used to limit the risk of negative side effects from non-personalized medicine by furthering personalized medicine through the accuracy and speedy testing of many different treatment options.
With continued and careful development of organoids, disease understanding and treatments, personalized medicine and so much more can continue to be improved, saving millions of lives.
————————————————————————————————-
Work Cited:
Agarwal, S., Saha, L., Sohani, A., Karri, U., Gor, R., & Ramalingam, S. (2024). The importance of niche in therapeutic strategies targeting normal and cancer stem cells. Cancer Stem Cells and Signaling Pathways, 15–33. https://doi.org/10.1016/b978-0-443-13212-4.00031-3
Bredenoord, A. L., Clevers, H., & Knoblich, J. A. (2017). Human tissues in a dish: The research and ethical implications of organoid technology. Science, 355(6322). https://doi.org/10.1126/science.aaf9414
Frans Schutgens, & Clevers, H. (2019). Human Organoids: Tools for Understanding Biology and Treating Diseases. Annual Review of Pathology Mechanisms of Disease, 15(1), 211–234. https://doi.org/10.1146/annurev-pathmechdis-012419-032611
Generation and Cultivation of 3D Cell Models | ibidi. (2024). Ibidi. https://ibidi.com/content/1037-generation-and-cultivation-of-3d-cell-models
Lancaster, M. A., & Huch, M. (2019). Disease modelling in human organoids. Disease Models & Mechanisms, 12(7). https://doi.org/10.1242/dmm.039347
O’Connell, L., & Winter, D. C. (2019). Organoids: Past Learning and Future Directions. Stem Cells and Development, 29(5), 281–289.https://doi.org/10.1089/scd.2019.0227
Schutgens, F., & Clevers, H. (2020). Human Organoids: Tools for Understanding Biology and Treating Diseases. Annual Review of Pathology: Mechanisms of Disease, 15(1), 211–234. https://doi.org/10.1146/annurev-pathmechdis-012419-032611
Simian, M., & Bissell, M. J. (2016). Organoids: A historical perspective of thinking in three dimensions. The Journal of Cell Biology, 216(1), 31–40. https://doi.org/10.1083/jcb.201610056
Sun, D., Gao, W., Hu, H., & Zhou, S. (2022). Why 90% of clinical drug development fails and how to improve it? Acta Pharmaceutica Sinica B, 12(7), 3049–3062. https://doi.org/10.1016/j.apsb.2022.02.002
Visvikis-Siest, S., Theodoridou, D., Kontoe, M.-S., Kumar, S., & Marschler, M. (2020). Milestones in Personalized Medicine: From the Ancient Time to Nowadays—the Provocation of COVID-19. Frontiers in Genetics, 11. https://doi.org/10.3389/fgene.2020.569175
Vlachogiannis, G., Hedayat, S., Vatsiou, A., Jamin, Y., Fernández-Mateos, J., Khan, K., Lampis, A., Eason, K., Huntingford, I., Burke, R., Rata, M., Koh, D.-M., Tunariu, N., Collins, D., Hulkki-Wilson, S., Ragulan, C., Spiteri, I., Moorcraft, S. Y., Chau, I., & Rao, S. (2018). Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science, 359(6378), 920–926. https://doi.org/10.1126/science.aao2774
Xu, H., Jiao, Y., Qin, S., Zhao, W., Chu, Q., & Wu, K. (2018). Organoid technology in disease modelling, drug development, personalized treatment and regeneration medicine. Experimental Hematology and Oncology, 7(1). https://doi.org/10.1186/s40164-018-0122-9


