Welcome back to our discussion on the Drake Equation and the Fermi Paradox! If you haven’t read part 1 already, it goes without saying that you should start there! This time around, we will be discussing what we know about habitable planets… or at least what we think we know. Strap in, friends. This is going to be a long one.
Before we proceed any further we need to introduce the concept of ‘filters’, i.e. factors that may inhibit the development of life and, ultimately, intelligent, detectable civilizations (IDCs) on a given planet. It is crucial to keep in mind that all proposed filters are not absolutes, rather they are speculative reasons why IDCs may be rare, thus resolving the Fermi Paradox. For convenience we can designate “Lesser” filters as those which eliminate 0% to 50% of all planets, “Minor” filters which eliminate 50% to 90%, “Major” filters which eliminate between 90% and 99.9999%, and finally “Great” filters which eliminate 99.9999% to 100% of planets. With this caveat in mind, we move on to our next parameter in the Drake Equation.
The Average Number of Habitable Planets per Star with Planets, ne
In order to determine what types of planets can support life, we first need to know what conditions are actually right for life, a difficult task given our current sample size of one. Of the eight planets in our solar system, only Earth is known to host any life at all. Though the emerging field of astrobiology is beginning to shed new light on the issues of whether or not life could have existed on Mars and Venus in the past or concurrently in the subsurface oceans of moons like Enceladus and Europa, the question remains open. Note that we are only looking for planets with the potential to support any life for this number, be it microbial or otherwise. We certainly do not expect microbes to start sending messages out into space, but the distinction between life and intelligent life is one that will be accounted for by subsequent numbers in our equation. This will allow us to save many of our potential filters for later and simply focus on habitability in all its possible forms. Sadly, those that remain are the ones we are most certain about.
We can begin to approach the problem by flipping it around, instead asking the reverse, “What conditions preclude life from developing?” If we focus exclusively on planets, our sample size has septupled since there are seven planets in our solar system that are known to not support life. Investigating the factors that prevented life from existing for a significant amount of time provides us with some starting filters which allow us to eliminate entire swaths of planets from the veritable zoo of those discovered.
Filter 1 – Location, location, location!
The clichéd real-estate adage provides us with our first filter. Since we are focusing solely on the Milky Way, we don’t need to worry about galaxy type. Our own existence is proof that our galaxy is among those that are right for hosting life, so we need not concern ourselves with filters like active galactic nuclei, galaxy mergers, etc. However many astronomers propose that a star system’s location within a galaxy is an important factor. There may very well be a so-called “habitable zone” on the galactic scale in addition to that found around the individual stars themselves. Close to the crowded galactic center a system is likely to be subjected to nearby supernovae, migrating black holes, asteroid/comet impacts, passing pulsars, and other devastating natural phenomena with the potential to sterilize entire planets. On the other hand, systems at the galaxy’s edge are unlikely to have enough nearby gas and dust to sufficiently drive the formation of stars and planets. The average stellar metallicity (how much of a star is composed of elements heavier than helium) declines as distance from the center increases, so stars near the galaxy’s edge are expected to have fewer rocky, potentially life-sustaining planets. As it turns out, this may not ultimately be the case as some data suggests rocky planets could be more common around lower-metallicity stars, but for now this will be our working assumption.
This limits the expected life-sustaining region of our galaxy to stars whose orbits lie from at least 1 to at most 10 kiloparsecs from the center. For reference the Milky Way has a radius of only about 15 kiloparsecs, and with a little data and some rough assumptions, one can calculate that only around 10% of all stars in our galaxy lie in this region. This number is still staggeringly large and includes, to first order, around 20-40 billion stars or so. Of those we can reasonably exclude the 10% of stars in our galaxy that are not on the main sequence. Given that the average number of planets per star may be as high as 10 on the extreme optimistic end, this leaves a maximum of about 360 billion planets to work with.
In addition to the galaxy, the stars themselves each have their own habitable zones, defined as the range of orbital distances within which liquid water could conceivably exist on the surface in large quantities. There are only three planets in our solar system that exist more or less within the sun’s habitable zone: Venus, Earth, and Mars. Of these three, Venus and Mars are each close to the edge of what can be considered the habitable zone, and given the concept’s speculative nature these boundaries could grow or shrink with new information. The most we can do right now is place an upper limit on this value of 6, based on research by Dr. Christa Van Laerhoven and her colleagues, though the actual number is almost certain to be lower. For simplicity, we can estimate the average number of planets in a star’s habitable zone to be 3. The size and location of the habitable zone is determined by the type of star. Large stars (O, B, and A types) account for only about 7% of all stars and tend to burn out too soon for complex life to really gain a foothold. It’s uncertain whether planets around smaller, red dwarf stars might host life. The data indicates the planets orbiting in their habitable zones may be tidally locked to the star just as the moon is to the Earth, with one side facing the star at all times. While this doesn’t preclude life from forming, it certainly makes things more difficult. Red dwarf stars may account for as much as 75% of the stars in our galaxy, so whether they should be included in the search for life is a crucial question that will hopefully be answered as more data becomes available. Nonetheless we have our next filter: of the main sequence stars, at most 93% of them are expected to be habitable.
There are without a doubt more particulars of planetary arrangement that come into play here, such as having a stable, mostly circular (low eccentricity) orbit, the role gas giants like Jupiter may play as cosmic vacuum cleaners, and more, but the only thing we know for sure regarding the rest of the Location Filter is that we just don’t know.
Filter 2 – Earth-Sized:
Planets that are too large tend to be gas or ice giants, with no solid surface and crushingly thick and turbulent atmospheres. Life could form here, but if it does, it’s likely the exception rather than the rule. At the same time, the cores of planets that are too small tend to cool too quickly and lose their magnetic fields and subsequently their atmosphere to stellar winds. Mars is a perfect example of this. Looking at the best current estimates, we can see that only around 1,237 of the 4,370 exoplanets discovered so far are “Earth-sized”, i.e. range from 0.8 to 2 Earth radii. We can be generous here and allow for the fact that exoplanet survey methods are biased towards larger planets by estimating that perhaps 40% of planets are Earth-sized.
Filter 3 – Atmospheres, Plate Tectonics, and Moons:
Being Earth-sized helps when holding onto an atmosphere, but this does us no good if it isn’t the right atmosphere, and we need look no further than our own solar system for the perfect illustration of this. Venus is 93% the mass of the Earth, yet its atmosphere is crushing at a choking 100 times the density of Earth’s. Unlike our own atmosphere which is about 70% Nitrogen, Venus’s atmosphere is more than 96% CO2, 30 times the CO2 concentration of our own atmosphere. This runaway greenhouse effect famously makes Venus the hottest planet in the solar system, with surface temperatures hot enough to melt lead. Unfortunately we have no real data for this yet as we lack the ability to reliably observe exoplanet atmospheres, though that is expected to change with the launch of the James Webb Space Telescope set to launch in October of 2021. For now, we will be generous and use our own solar system as the model by assuming that 50% Earth-sized planets do not have the right atmosphere for life.
Two other factors that make Earth, well, Earth-like are plate tectonics and our Moon. Plate tectonics play a crucial role in cycling numerous elements, including carbon and phosphorus, in and out of the environment. It’s thought that Earth’s water and the theoretical collision with a Mars-sized planet which formed the moon are two of the main factors that got plate tectonics going. Venus, which lacks both, shows no signs of tectonic activity, past or present, even though its core is known to still be molten and active. The Moon is somewhat of an anomaly in its own right. Of all the planetary moons in the solar system, it is by far the largest relative to the size of its host planet. Its existence helps to stabilize Earth’s axial tilt and over billions of years, through a quirk of angular momentum conservation, gradually slowed our planet’s initially brisk rotation to the 24 hour day/night cycle we experience today. It also generates tides and thus tidal pools, which some theories for abiogenesis claim provided the conditions for the origin of life on the early Earth. Again, we will be generous here and presume that 1 in 2 Earth-sized planets in the habitable zone of their star are actually Earth-like.
Filter 4 – Phosphorus Scarcity:
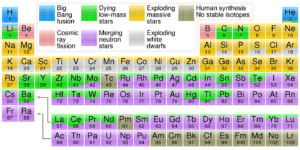
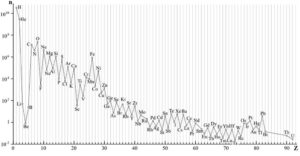
A recent study of two supernovae remnants, the Crab Nebula and Cassiopeia A, by Dr. Jane Greaves of Cardiff University and her team suggests that phosphorus may be produced in vastly different quantities during supernova nucleosynthesis. It’s somewhat common knowledge, even among the general public, that the Big Bang produced only Hydrogen, Helium, and a possibly a small amount of Lithium, and that all elements higher on the periodic table were produced in the cores of stars as byproducts of nuclear fusion, a process known as hydrostatic burning, but this is only half of the story. Stellar nucleosynthesis can only account for the production of a specific set of elements, most of which have an even number of protons due to their fusion being reliant on Helium and none of which are heavier than Zinc (element 30). All elements with more than 30 protons can only be naturally produced by what happens after stellar nucleosynthesis ends, in the explosive deaths of stars and the violent collisions of their remnants. As the star collapses infalling matter piles up and is “bounced” back out, producing large numbers of neutrons and sending shockwaves through the star’s layers which quickly fuse lighter elements into heavier ones through the process of rapid neutron capture, known as the r-process. In fact the majority of the elements trace their origins to events beyond the cores of main sequence stars as can be seen in the chart below. All of this results in massive discrepancies in universal abundances between elements, shown in the second image. Taking particular note of what the data shows regarding the six elements considered most critical for life: Hydrogen, Carbon, Nitrogen, Oxygen, Phosphorus, and Sulfur. Phosphorus in particular is required for the metabolic processes of all known cells (via ATP) and forms the backbone of both DNA and RNA, yet it is by far the least common of the six.
(By Cmglee – Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=31761437, based on data by Jennifer Johnson at Ohio State University)
The most likely production process involves a silicon-30 isotope capturing a neutron during the supernova, briefly becoming unstable silicon-31 before one of its neutrons emits an electron and an antineutrino to decay into a proton, transmuting the atom into phosphorus-31, the element’s only stable isotope. The problem is that silicon-30 is not the most abundant Si isotope, accounting for only about 3.1% of all naturally-occurring Silicon, and its cross-section for neutron capture is quite small, meaning a neutron has to hit it just right to get absorbed. Supernovae are not evenly distributed around the galaxy, and thus we already expect that phosphorus isn’t either. Should it be the case that Phosphorus is not a ubiquitous byproduct of supernova nucleosynthesis, the element will only be rarer and more unevenly distributed than previously thought. There could very well be entire swaths of the galaxy with virtually none at all.
While it might be possible for life to develop that does not rely on phosphorus, the chances of that actually happening are probably slim, and such life would be the exception rather than the rule. We ourselves are composed of approximately 1% phosphorus, the planet’s crust is about 0.1% phosphorus, and the entire solar system is roughly 0.0007% phosphorus by mass. On the scale of the entire universe, phosphorus only accounts for about three of every ten million atoms, or 0.00003%, and would have been even lower than that when our solar system began to form 5 billion years ago. Naturally, older star systems are expected to have less phosphorus on average as fewer phosphorus-producing supernovae would have occurred prior to their formation. Even our apparent relative abundance of phosphorus is so low that it presents a problem for scientists seeking to tease out the origins of life. Phosphorus also tends to bind into molecules that are heavier and thus would have sunk towards the core during the planet’s formation, though the collision which we think formed our Moon may have played a role here in dredging some up from the Earth’s interior. Its common compounds also tend to be insoluble in water which is problematic since life processes are pretty much predicated entirely on water solubility. Unlike the Earth’s carbon cycle which is relatively quick on geological timescales and involves the atmosphere as well as the planet’s interior, the phosphorus cycle is one of the slowest and independent of the atmosphere, so biologically-available Phosphorus is only refreshed by tectonic activity. That life should rely so ubiquitously on phosphorus despite its apparent scarcity tells us either that phosphorus-based life is easier to form than non-phosphorus-based life or that life based on phosphorus has an evolutionary advantage over life that is not. Possibly even both. Water is quite common in the universe, and so it may be that Phosphorus is what makes Earth truly unique among the planetary zoo.
Until there is conclusive evidence one way or another, we can take this potential filter with a grain of salt and estimate that only 50% of planets have a sufficient amount of Phosphorus present on the surface. We can now combine all our filters into one to calculate a final value for ne and find that several Lesser and Minor Filters have combined to form one Major Filter. With less optimistic estimates, such as excluding red dwarf stars, and a few more assumptions and factors accounted for, this has the potential to reach Great Filter status and serves as the foundation for the Rare Earth Hypothesis.
ne, Optimistic = 3 x 0.1 x 0.9 x 0.93 x 0.4 x 0.5 x 0.5 x 0.5 = 0.0125
ne, Rare Earth < 0.000001
For now, this is where we will leave the second half of our discussion of Drake’s equation. We can take all the factors we’ve discussed so far, multiply them together, and multiply this value by the length of time life has existed on Earth (3.5 billion years) to get a somewhat crude estimate for the number of planets in the galaxy which might contain life in any form.
NOptimistic = 131,250,000
NRare Earth < 10500
When compared against the hundred billion or so planets that are estimated to reside within the Milky Way, even our deliberately generous estimate indicates that only around 0.13% of planets harbor any life at all. If we apply the Rare Earth filter for ne, then only about 0.00001% of planets in the Milky Way are safe havens for life. These numbers will only diminish as we start accounting specifically for intelligent life in subsequent factors of the Drake Equation. After all, the Fermi Paradox is specifically about why we haven’t heard from any other intelligent life, not merely the chances that extraterrestrial life in general exists.
Next time we’ll be discussing that second half of the Drake Equation and the implications further proposed filters have for our chances of detecting intelligent life within the Milky Way. Unfortunately, the last three factors rely heavily on speculation in the absence of virtually any reliable data, and we will delve deep into some of that speculation in part 3. In part 4 we’ll discuss the final factor of the equation and run some example calculations before finally delving into what the Drake Equation actually is. In the meantime, if you’d like to try your hand at estimating the number of inhabited planets or intelligent civilizations in the universe, you can use this handy link for calculating the Drake Equation.