PROFESSOR: Hello class! Welcome to “Advanced Application of Math and Logic in Natural Philosophy” class. Look to your left and look to your right. One out of five in this room will not pass the class.
STUDENT A: Sir, but there are only four of us.
PROFESSOR: (ignoring the student) Ahem. During our first lecture, we will exercise the use of logic and thought experiments to unveil one of the greatest mysteries in the history of science. Even Aristitotle struggled solving this mystery. For centuries many scientists attempted to solve but failed. (drum roll) Why does hot water freeze faster than cold water?
STUDENTS B & C: (smile nervously not knowing whether he is joking or not)
STUDENT A: But sir, hot water needs to be cooled down before it can freeze. So shouldn’t cold water freeze faster?
PROFESSOR: Good point. However, that is not always true. It’s called the (writing on the board) Mpemba effect. Can anyone guess why? It’s okay even if you are wrong. Don’t be shy – that is why I am here.
STUDENTS A, B, C, & D: (silence)
(Satisfied with his authority and the full attention of his students, the professor continues)
PROFESSOR: Actually, experiments showed that hot water will cool faster than water at 60°C. But this is not the case with water below 60°C. Mpemba effect is a combination of multiple factors. Now let’s figure out each of them together. First, tell me what are the characteristics of hot water?
STUDENT B: It has higher average kinetic energy.
The water molecules move faster, which means that they occupy greater volume, similar to how an air balloon expands at higher temperature.
STUDENT C: Wait, that means that if cold water and hot water have the same volume, the mass of hot water will be less. This is because the evaporation is taking place and dissolved gases in hot water escape at a higher rate.
PROFESSOR: Great answers. Water is most dense at 4 degrees Celsius and its density changes according to its temperature. Yes, so 1L of water does not always hold the same amount of mass. 1L of hot water will weigh less compared 1L of water near 4°C. However, the mass difference is slim even when we considering the evaporation of water molecule. So there has to be another factor that contributes to Mpemba effect.
STUDENT: But sir? What about supercooling of water and nucleation? Doesn’t lack of impurities and gases in water lead to supercooling, in which water forms crystal ice at temperatures below 0°C? It has been observed that water can remain liquid at -40°C. If hot water doesn’t have dissolved gas, shouldn’t it be hard to make it freeze?
PROFESSOR: (as if expected) Good point. Except Mpemba effect has been observed in both hot water and cold water removed of dissolved gases. Dissolved gas does not seem to contribute to the process of freezing. You could, however, make beautiful and clear crystal ice by removing dissolved gas before freezing. So what else? What else does hot water do?
STUDENT C: It evaporates faster than cold water..?
PROFESSOR: Yes, the water evaporates faster. But why? And so what?
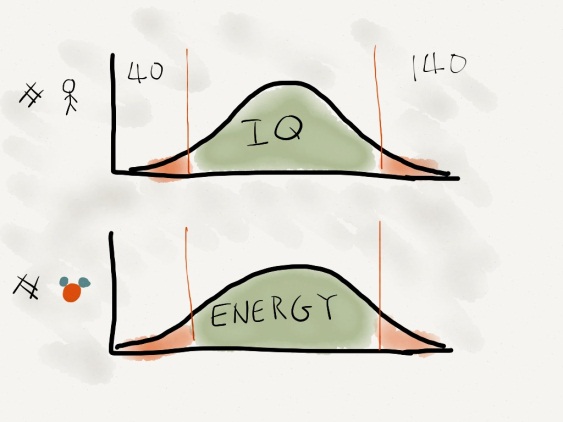
STUDENT B: Well umm… the temperature of water is the average kinetic energy. At any time, liquid water has molecules with a range of energies. Although most of them have the average kinetic energy of the system, very few have enough energy to escape intermolecular forces such as hydrogen bonds and dipole-dipole attractions. If we were to look at a graph of number of molecules versus energy, it would look similar to a current IQ graph, in which the majority of the population has an IQ of 100 and few have over 140 and under 40.
STUDENT A: (towards student B) Hydrogen bond and dipole-dipole forces are too weak to contain water molecules in liquid form. It’s actually the atmospheric pressure doing all the work.
PROFESSOR: Please let him continue.
STUDENT B: So molecules with higher energy escape, lowering the average energy within the system, and this is evaporative cooling.
STUDENT C: Oh! So the hot water is not only being cooled through evaporative cooling but also from heat conduction and convection inside the freezer.
PROFESSOR: Good answers class. But that is not enough. According to Dr. Takamasa Tkahashi at St. Norbert College, it takes 540 calories to vaporize one gram of water. At above 80°C, the rate of cooling by evaporization is high as vapor draws massive energy from water left behind. To give you an idea how much 540 calories worth, it takes 100 calories to heat up 1 gram of water to 100°C from 0°C.
STUDENT A: (excited) 1 Calorie is the energy required to raise 1 gram of water 1°C! In fact, scientists measured food calories by burning them and heating up water back in old days.
STUDENT D: Wow…is that why greasy foods have high calories?
PROFESSOR: If we neglect heat transfer through radiation or conduction, water will freeze on a desert purely from evaporation. Interesting, isn’t it? We can use zeolites which facilitate evaporation through adsorption to make ice as well. A team of engineers at Rice University built a prototype of ice machine that uses nothing but zeolites and vacuum pump.
STUDENT D: (perplexed) But professor, when hot water reaches the temperature of the cold water, don’t both of them have the same evaporation rate?
PROFESSOR: (impressed and amused) Very good point. I might actually consider allowing one A+ in this class. To answer your question, let’s first consider extreme cases to facilitate our thought experiments. Imagine placing an ice cube onto a very hot plate. What happens?
STUDENT A: (desperate to not embarrass himself) Uh…The ice melts immediately? And while some water molecules evaporate, others boil around the remaining ice cube, making noise from gases and vapor escaping. Noise is also another form of energy transfer.
PROFESSOR: Yes but melting the process does not happen instantly. While the ice is melting, what is the average kinetic energy? It is somewhere in between that of boiling water and ice.
Now, we learned that the energy of liquid water is not perfectly uniform. The faster the change of temperature is applied the greater inconsistency of the energy. Hence, hot water being cooled down to cold temperature has a higher evaporation rate than cold water that remained cold for a prolonged period of time. I think you’ll need to elaborate on this one…
STUDENT A: Also, Newton’s law of cooling dictates that hot water will cool down faster regardless of the evaporation.
PROFESSOR: Yes, of course, faster moving molecules will bounce off of each other more frequently, giving and losing energy through these interactions.
STUDENT D: So that means… initially, cold water has a more uniform temperature. But when hot water is cooled, although the average temperature is cold, there are greater discrepancies of temperature among molecules?
STUDENT A: (being impatient) Yes, and that is why the hot water will be more likely to have molecules with higher energy, which escapes and cools the liquid faster.
PROFESSOR: Like washing a car with hot water during winter, evaporative cooling is more evident with larger surface area. So what else? Does anyone else have any more idea?
STUDENT C: Well… this may seem stupid, but what if Mpemba effect is caused by other effects such as the environment?
PROFESSOR: (intrigued) What do you mean?
STUDENT C: The freezer keeps the temperature below zero all the time. But when the temperature increases, cold air is introduced in order to keep the temperature constant. So if we place hot water inside the freezer, and due to heat conduction, radiation, and evaporation, the internal temperature increases, it will trigger the freezer to blow cold air. You need to elaborate on this… And thus, hot water will freeze faster.
PROFESSOR: Hmm…I like your creative way of thinking.
STUDENT B: Also, freezers have ice dusts inside. And hot water may melt these ice particles within the compartment at contact points of the bowl and the surface. Since water is dense and thus conducts heat faster, hot water will cool down at higher rate. The effect is similar to how ice water cools beer bottles faster than ice cubes.
PROFESSOR: Excellent point! Water is 829 times denser and transfers heat 25 times faster than air. Hot bowl of water sitting on slightly melted ice will definitely cool faster than cold bowl of water sitting on ice dust. Water covers more surface area as well. But aren’t you slightly underage to know how to cool beer bottles?
STUDENT A: (hungry for attention) Sir, what about infrared radiation? We know hot objects emit electromagnetic radiation and thus release energy at a faster pace. A combination of this and everything we discussed today will make hot water cool faster.
PROFESSOR: Now, do you guys remember how water changes its density? Due to heat conduction at contact points, a bowl of water will be colder at the bottom. Let’s consider a cold bowl of water. Cold water near freezing point at the bottom of the bowl will have lower density than water at near 4°C on top. Low density water will rise up switching position with water near 4°C. Neither upper nor lower part of water will freeze until that 4°C temperature is brought to 0°C.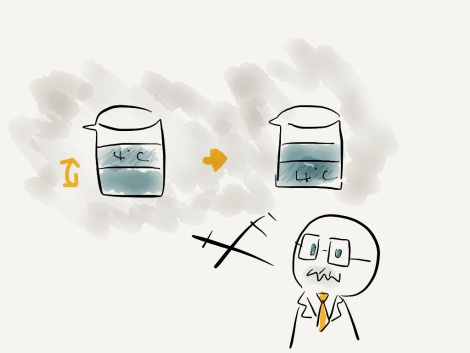
Now what about hot bowl of water? Hot water at the top has lower density than cold water at the bottom. Thus, cold water near freezing point won’t rise up. This means the water at the bottom can freeze before hot water is cooled to 4°C. This effect will freeze hot water faster.
STUDENT C: Whoa… there seem to be a lot of factors that play in Mpemba effect. We discussed mass difference, evaporation rate, freezer compartment effects such as more frequent cold air and melting ice at contact points, and how density can slow or speed up the freezing rate. I don’t think we can come up with anymore explanations.
PROFESSOR: Aha but we are not done! There are more. What are we forgetting class?
STUDENT D: (as if he had more than enough to learn) Not a clue sir…
PROFESSOR: (excited) The internal air pressure of the freezer compartment~!
STUDENT D: But how..?
PROFESSOR: (showing a video clip on a TV) Do you guys watch a BBC TV series called “Bang Goes the Theory”?
STUDENT C: Oh my gosh! I absolutely love that show!!
PROFESSOR: Let’s look at season 2 episode 4. In this episode Dr. Yan shows that when you quickly open a bottle there’s a cloud of white air at the opening. This effect is the opposite of how diesel engine ignites a spark by compressing air. (Professor demonstrates by opening a beer bottle). The air between the liquid and the cap is quickly expanded and loses energy. At this very instant, the temperature drops to around -30°C and this white gas you see is the condensed water molecules in the air.
STUDENT A, B, C&D: (intrigued) wow…
PROFESSOR: As the ideal gas law states, hot air has lower density than cold air under equal pressure and volume. If hot water and cold water were placed in a separate freezer compartments, the one with hot water will have less air molecules inside.
STUDENT D: But professor, why?
PROFESSOR: Because hot water will heat up the air inside the compartment. Of course, the longer you wait before closing the door, the more evident the effect would be. You all saw how quickly air cooled down when I opened the beer bottle. (Takes couple of sips of beer). You can create the same effect by opening the freezer compartment. Now when you open both hot and cold water compartments, the one with hot water will experience greater decrease in temperature; air with lower density will lose more energy when expanded at the same volume as air with higher density. This gives the last minute boost of freezing air to hot water compartment.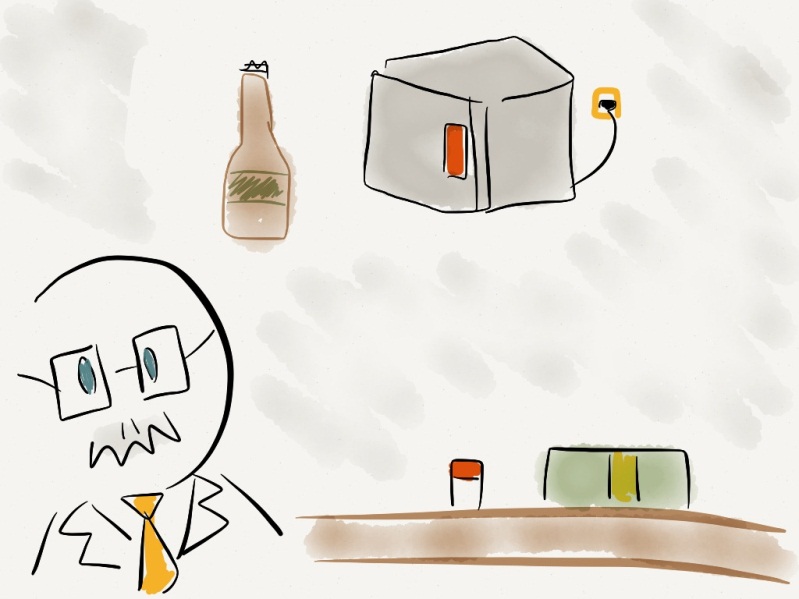
But even if you don’t open the freezer, lower pressure will facilitate freezing. Can anyone guess why?
STUDENT A: As we can see on the phase diagram, the freezing point of water is higher at lower pressure. Hence, by lowering the pressure, hot water will freeze at higher temperature than cold water.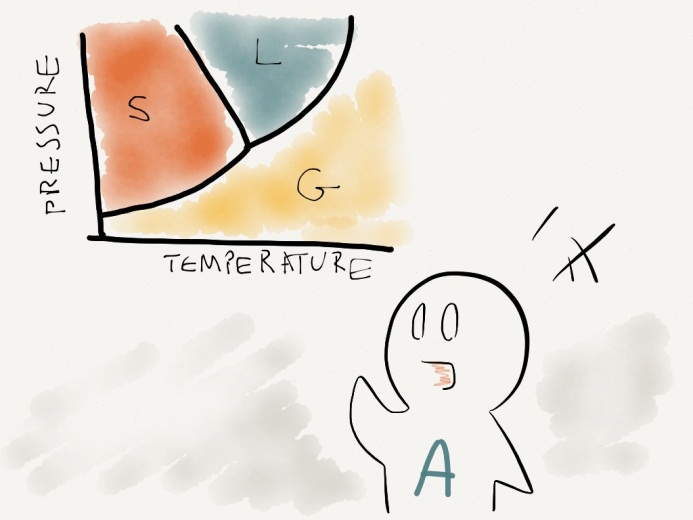
PROFESSOR: Excellent! Although the boiling point is lower at low pressure, freezing point is higher. Since ice occupies greater volume than liquid water, applying pressure to ice cubes will turn solid into liquid. Not only does lowering pressure increase evaporation rate, it helps to crystalize ice!
STUDENT C: I see… Mpemba effect isn’t magic. It’s science.!
STUDENT D: (frustrated) But professor, none of these answers answer any questions at all! Why must water evaporate? How come its temperature is not uniform? How come its density changes with the temperature? Why all this?
PROFESSOR: This class is a natural philosophy class for a reason. Science is a study and observation of the universe, finding pattern in its chaotic behavior and thereby hypothesizing the answer with logic. And we test our hypotheses to come up with a prediction. We, scientists, ask the question ‘why?’ and dwell on solving the mystery. But the answer we find is the answer to the question ‘how?’ We observe the mechanism and apply it, but we still don’t know why the universe behaves in the way it does. For example, why does Earth create gravitational attraction? Einstein explained that it is because mass curves the space. But why does mass create a distortion of space and why does the distortion cause attractive forces? We don’t know. All we know is how mass creates gravity
Class, I encourage all of you to pursue knowledge with an inquisitive mind. Scientific knowledge is what shapes our future.
Class dismissed. Write me 50 pages on Mpemba effect by tomorrow. It should be easy now that we discussed it in class.