It’s easy to forget that some of the minerals found in the Earth can be extremely dangerous, despite the beautiful forms they can take. These minerals can prove to be toxic and potentially deadly if mishandled. So I present to you seven minerals toxic minerals, so you can identify them if you ever (have the misfortune to) come across them:
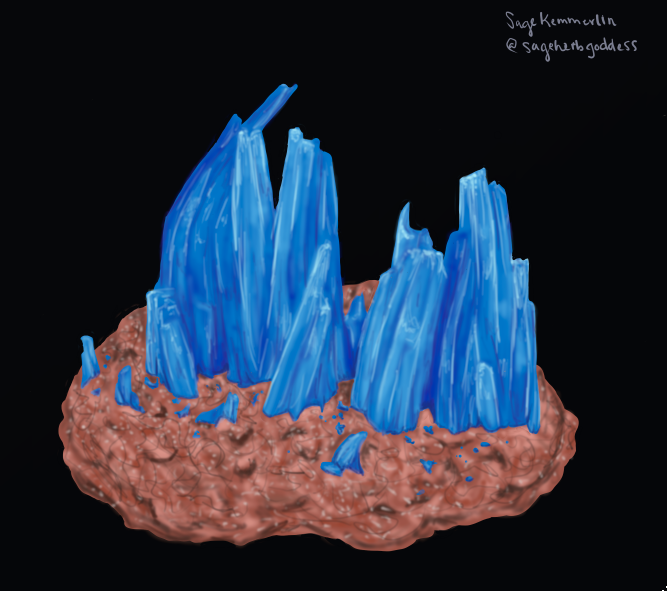
Chalcanthite (CuSO4 • 5H2O):
Image Credit: Sage Kemmerlin
The stunningly blue mineral is a natural, water-soluble copper sulfate. It is commonly found in copper deposits in arid regions. If chalcanthite touches water it easily dissolves and become hazardous because of the copper it contains. If copper is ingested, the effects of this poisoning are gastrointestinal distress, destruction of red blood cells and liver damage (sounds fun right?). If the copper poisoning is severe, these symptoms can lead to death. Chalcanthite should never be taste tested or placed in natural waters. [1][2]
Hutchinsonite (TIPbAs5S9):
Image Credit: Sage Kemmerlin
Hutchinsonite is commonly found in ore deposits in the mountainous regions of Europe. Hutchinsonite is a mineral formed with a deadly mixture of arsenic, lead, and thallium; all three elements are toxic to humans. Thallium salts have been used in rat poison and insecticides so that alone should tell you not to mess with this mineral. If poisoned with thallium, you only have 72 hours to start treatment to avoid risk of permanent neurological damage. So maybe don’t lick it. [3]
Orpiment (As2S3):
Image Credit: Sage Kemmerlin
This orange-yellow, glistening mineral is made up of arsenic and sulfur and commonly grow near hydrothermal vents, fumaroles, and hot springs. If allowed to oxidize, the arsenic powder can be deadly to humans due to its neurotoxic and carcinogenic properties. Orpiment has a powerful garlic scent due to the arsenic. If you see a pretty orange mineral that smells like garlic, make like a vampire, and get out of there. [4]
Galena (PbS):
Image Credit: Sage Kemmerlin
Galena is the primary ore mineral of lead with a signature cubic structure. Any inhalation or ingestion of Galena’s dust will likely result in lead poisoning. Lead can effect every organ system in the human body, but the nervous system is most sensitive to lead’s toxicity sometimes causing ataxia, comas, convulsions, even death. So if you want to avoid maladies such as ataxia and death, it’s best to not lick Galena or inhale any of its dust. [5][6]
Tobernite (Cu(UO2)2(PO4)2•12H2O):
Image Credit: Sage Kemmerlin
Tobernite is a stunningly green prism shaped mineral, found in granitic rocks. Oh and just as a side note, torbernite is radioactive due to the uranium it contains. So if you prefer not to get cancer, avoid prolonged exposure and never ever ingest or inhale it. [7]
Cinnabar (HgS):
Image Credit: Sage Kemmerlin
This brilliantly red mineral is the main ore of mercury. It forms near volcanoes and sulfur deposits. It has historically been used as a coloring agent. Mercury is toxic to humans and mercury poisoning can cause tremors, muscle atrophy, and in extreme cases, respiratory failure and death. As before, don’t ingest or inhale any cinnabar. Don’t heat it in an unventilated environment, because it will emit toxic fumes. So maybe don’t color your pottery with cinnabar (it’s not a good idea). [8][9]
References:
- http://www.mindat.org/min-959.html
- http://des.nh.gov/organization/commissioner/pip/factsheets/ard/documents/ard-ehp-9.pdf
- http://www.mindat.org/min-1954.html
- http://www.mindat.org/min-3021.html
- http://www.mindat.org/min-1641.html
- https://www.atsdr.cdc.gov/csem/csem.asp?csem=7&po=10
- http://www.mindat.org/min-3997.html
- http://www.mindat.org/min-1052.html
- http://www.medicinenet.com/mercury_poisoning/article.htm